So thermodynamically, the reaction can occur (in principle) - however, it is so slow that it cannot be observed effectively. However, this gas mixture is stable at ambient conditions. As an example, we have calculated in section 6.7.6.2 that the reaction of hydrogen gas and chlorine to hydrogen chloride has a negative value Δ G f 0. 
Δ G f 0 therefore only refers to the thermodynamic stability of a product that is equivalent to considering the equilibrium of a reaction. 
Reactions with values Δ G f 0 ≪ 0 will tend to run to completion while reactions with values Δ G f 0 ≫ 0 will yield almost exclusively the educts. Also, Δ G f 0 gives no information about the speed of the reaction, but merely about the degree of completion. As the changes in entropy (especially if the formation or consumption of a gas is involved) may be significant, exothermic reactions may still yield a positive Gibbs free energy and thus, will not occur spontaneously. It is important to note that merely judging a reaction exothermic and endothermic by looking at the enthalpy changes alone is not a sufficient manner in which to judge if a reaction will effectively occur. 
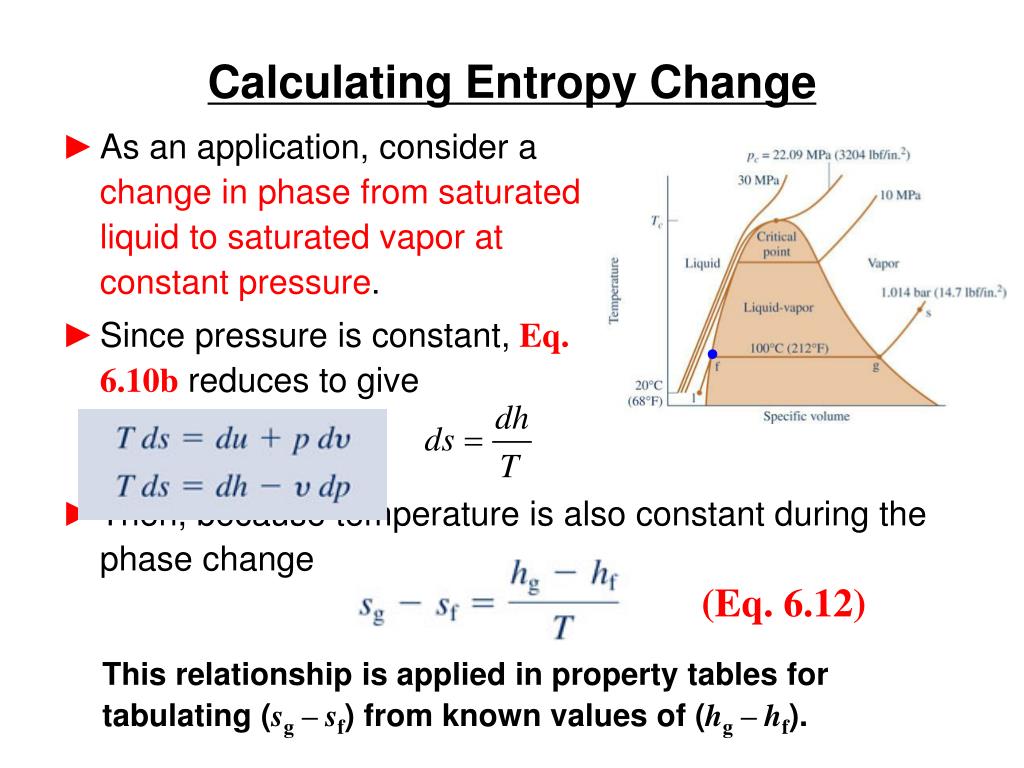
Δ G f 0 = 0: the reaction is in equilibrium usually values between − 5 kJ mol −1 and + 5 kJ mol −1 are considered to be equivalent to 0 Δ G f 0 0: the reaction will not occur spontaneously if the entropy is increased (by external heating), the Gibbs free energy may become sufficiently negative for the reaction to occur Thus adding up the entropy contribution and the enthalpy change, three cases must be considered. However, in chemical reactions, the entropy change due to the reaction must also be considered (see Eq. We now present this derivation and encourage the reader to follow it through, and in the process, gain a deeper appreciation for the Second Law of Thermodynamics.įor an exothermic reaction ∆ H 0 and therefore the entropy change of the ambiance is negative as heat is transferred into the system. This approach, which relies upon a fundamental mathematical theorem known as the Carathéodory theorem, is a most intellectually stimulating and satisfying exercise that leads to a much deeper understanding of the Second Law and the significance of entropy. However, it is possible to obtain equations (2.38) and (2.41) and show the equivalence of the thermodynamic and Absolute temperature scales without relying upon such idealized devices. Furthermore, in the next chapter we will apply the Carnot cycle while using an ideal gas as the working fluid to show that the thermodynamic (Kelvin) temperature scale and the ideal gas (Absolute) temperature scale are the same. In summary, we have seen how the introduction of the idealized Carnot engine leads to the definition of the thermodynamic temperature, an equation for calculating an entropy change from the flow of heat in a reversible process, and to the mathematical formulation of the Second Law in terms of entropy changes. Today, the Second Law, as applied to chemical systems, is firmly associated with the concept of entropy as expressed in the 1865 statement of Clausius and given mathematically by equation (2.41). Similarly, Helmholtz Free Energy F= U− TS (where U is internal energy) is useful in situations where volume and temperature T are constant, but it is not a fundamental quantity either. This makes Gibbs Free Energy extremely valuable for chemists, but it should not obscure the fact that entropy is the fundamental physical quantity and that Gibbs Free Energy is a derived quantity of no independent physical importance. Tables of Gibbs Free Energies of common substances are readily available, and such a table is all one needs to determine how entropy changes when an event occurs under conditions of constant pressure and temperature. Adding these two entropy changes together yields a total entropy change of Δ H/ T−Δ S=(1/ T)(Δ H− TΔ S)=(1/ T)Δ G, where Δ G is known as the Gibbs Free Energy. Under conditions of constant pressure and temperature, the former is the energy released by the system into its environment (traditionally represented by −Δ H) times 1/ T, and the latter is Δ S. To calculate the entropy change when an event occurs, the resulting entropy change of the system's surroundings must be added to the entropy change undergone by the system itself. Lozada, in Encyclopedia of Energy, 2004 5.4 Entropy and Free Energy 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
